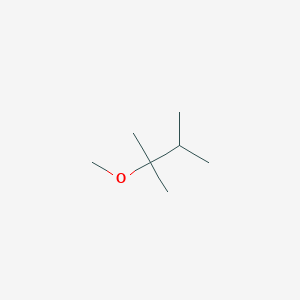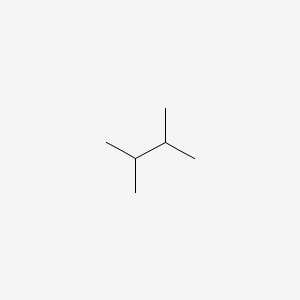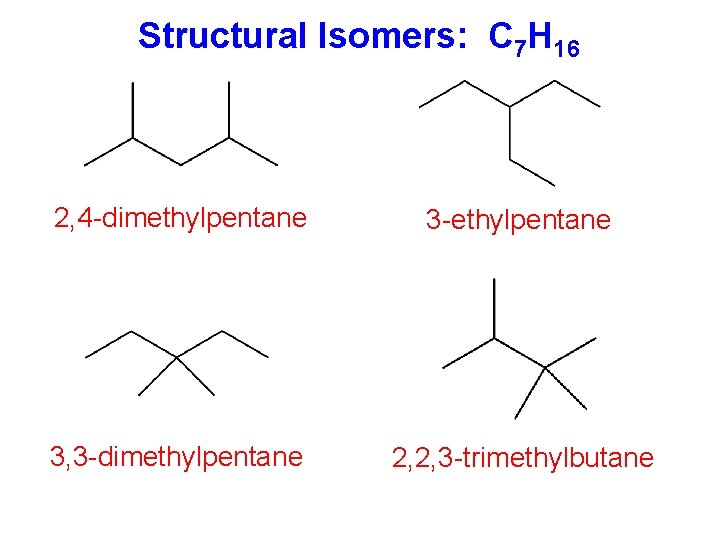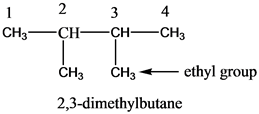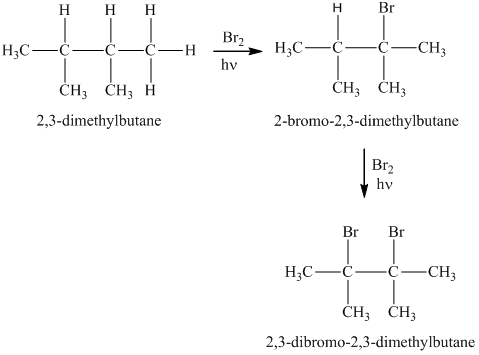
Solved: Chapter 4 Problem 24P Solution | Organic Chemistry Plus Masteringchemistry With Etext -- Access Card Package 9th Edition | Chegg.com
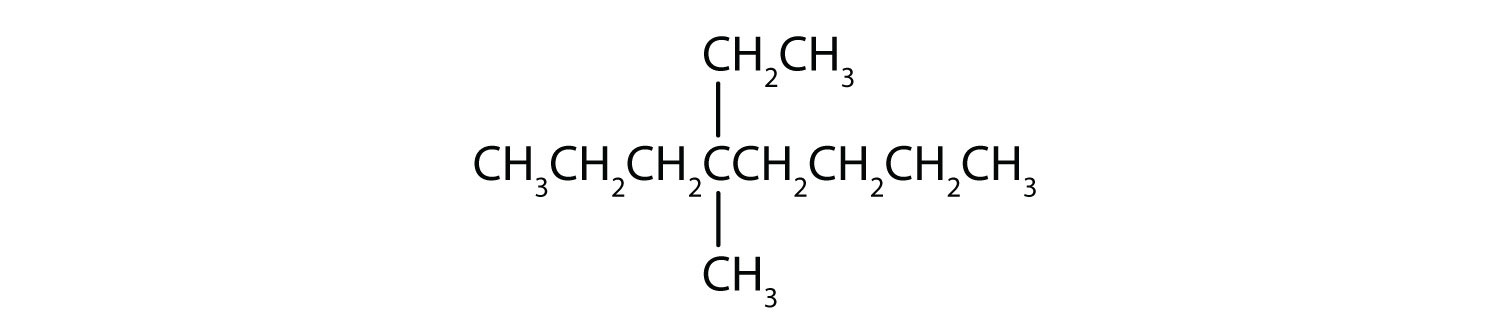
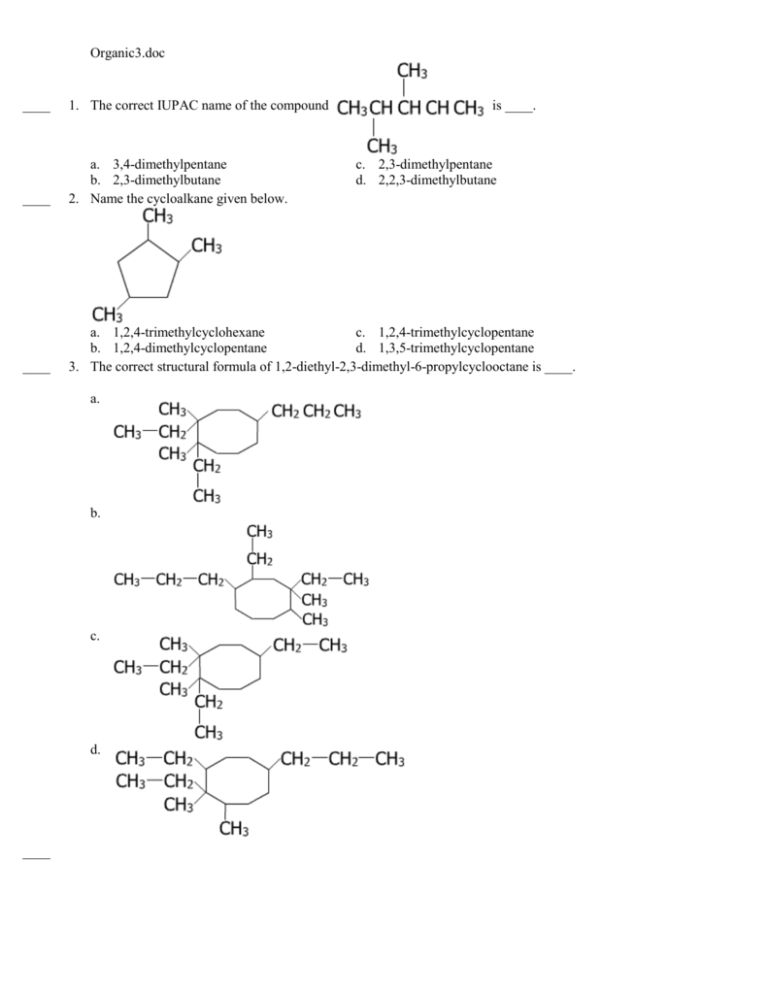
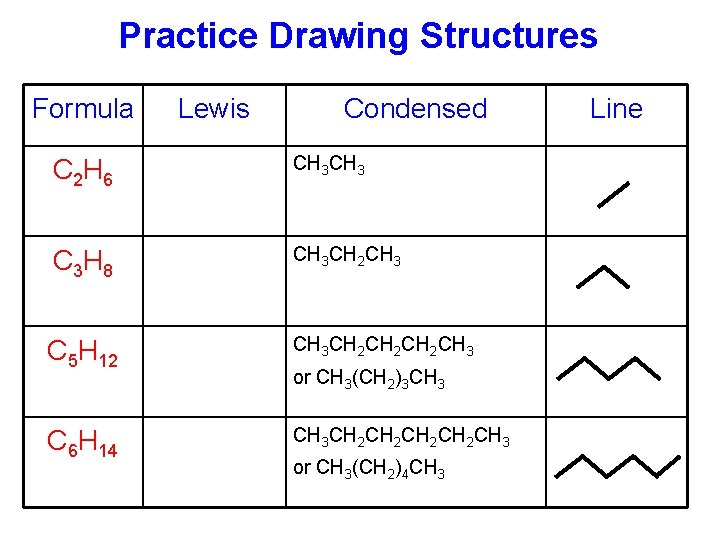
Chapter 1 - Organic Chemistry Review / Hydrocarbons - CHE 120 - Introduction to Organic Chemistry - Textbook - LibGuides at Hostos Community College Library

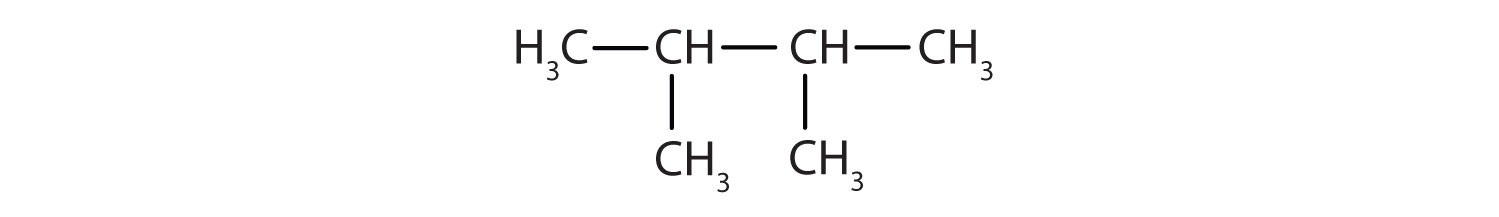
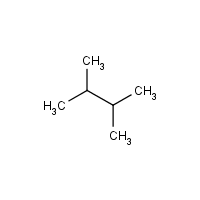
Draw the condensed and skeletal structure of 2, 3-dimethylbutane. What are the geometries about each of the carbon atoms? | Study.com

Out of 2,3-dimethylbutane and 2,2-dimethylbutane which has a higher boiling point And why - Chemistry - Hydrocarbons - 9885928 | Meritnation.com
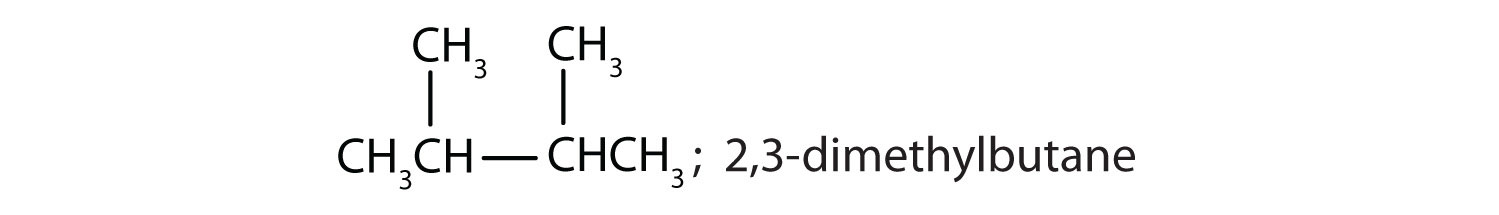
Chapter 1 - Organic Chemistry Review / Hydrocarbons - CHE 120 - Introduction to Organic Chemistry - Textbook - LibGuides at Hostos Community College Library
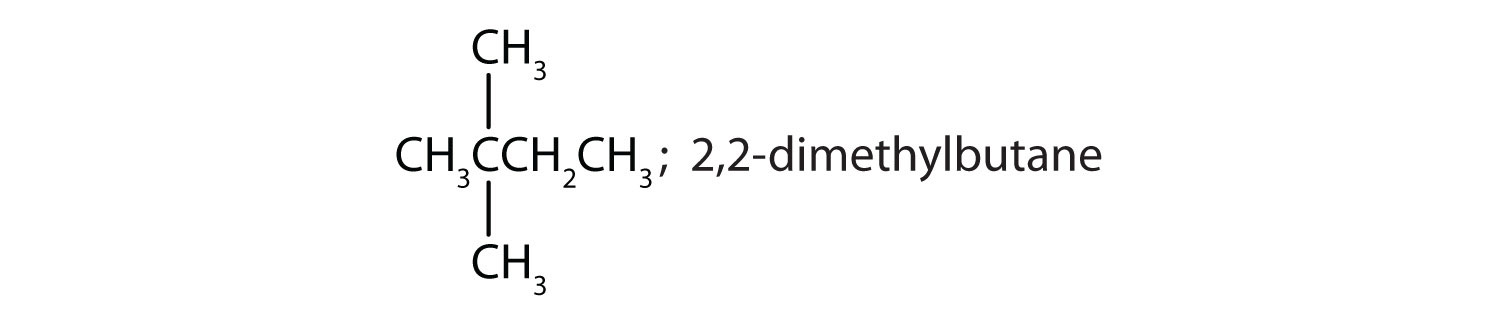
Chapter 1 - Organic Chemistry Review / Hydrocarbons - CHE 120 - Introduction to Organic Chemistry - Textbook - LibGuides at Hostos Community College Library

2R)-1,3-dichloro-2,3-dimethylbutane Formula - C6H12Cl2 - Over 100 million chemical compounds | Mol-Instincts